ProductionĪluminum is produced from bauxite through a series of processes. Being highly versatile, it is used in the transportation (automobiles, aircrafts, etc.) and construction (exterior siding, structural use) sectors, packaging, furniture, cooking utensils, and in electrical and electronics industries. ApplicationsĪluminum is almost always alloyed. Almost all metallic aluminum is produced from the ore bauxite which forms under wet tropical or subtropical climates. Although a common and widespread element, not all aluminum minerals are economically viable sources of the metal. It is almost never found in the elemental state. For this reason, aluminum is primarily found in the Earth's crust as oxides or silicates. In nature it reacts rapidly with oxygen to form oxides that become bound into rocks. Overall, the Earth is about 1.59% aluminum by mass. It is also relatively cheap, highly conductive, and resists corrosion. Aluminum can be drawn into wire or rolled into thin foil, is non-toxic, non-adsorptive, splinter proof, and very low in density. CharacteristicsĪluminum visually resembles silver and is soft, non-magnetic and ductile with excellent thermal and electrical conductivity. The first industrial large-scale production method, named the Hall–Héroult Process, was developed in 1886. 
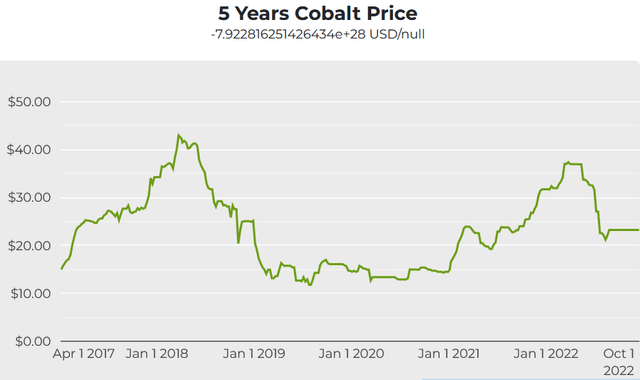
Since early methods could not yield great amounts of aluminum, its cost was more than that of gold. The first successful attempt, however, wasn't completed until 1824. Early Industrial ProductionĪttempts to produce aluminum metal date back to 1760. During the 20th century, aluminum became an indispensable part of everyday life as an essential component of housewares, and later as a much-sought civil engineering material. After the crusades, alum became an indispensable good in the European fabric industry, being imported from the eastern Mediterranean until the mid-15th century. The first recorded use of alum dates back to the 5th century BCE where it was used for city defense and dyeing mordant. The history of aluminum is largely shaped by the use of alum, which are chemical compounds of hydrated double salts containing aluminum. Today it is the second-most produced metal globally. Production has climbed steeply over the past two decades, particularly in China. Expressed as the symbol Al, this element has an atomic number of 13 and a density lower than most common metals. maybe, maybe not.Aluminum is the second-most produced metal globally and the most important non-ferrous metal.Īluminum, also called aluminium, is the most abundant metallic element in the Earth's crust. Our supplier, based in China, gives assurances that the origin of their metal is domestic. Iron, chromium and nickel end up being these stand-ins which end up yielding some form or other of “cobalt steel”.Įven though when all is said and done cobalt steel is usually 95% or more of these filler ingredients it is still a prized alloy for making tools out of especially those where you need a minimum of wear and tear.Īlso unfortunate for cobalt is that some 90% of the world’s supply is locked up dead in the middle of Africa’s least politically stable regions meaning that every ounce extracted from the earth comes with a tab in blood, sweat and tears. This puts the metal tinkerer in the ironic position of needing to create cobalt alloys not to improve their characteristics but rather to save on costs by diluting in other ingredients that aren’t so expensive. Pure cobalt, however, is almost a dream metal all to its own combining the desirable properties of being very hard, non-toxic and highly resistant to corrosion. Usually you need an alloy made up of several metals to achieve a recipe that checks all the marks for the ‘ideal’ metal for any given project.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
